What is Upgraded CGT test?
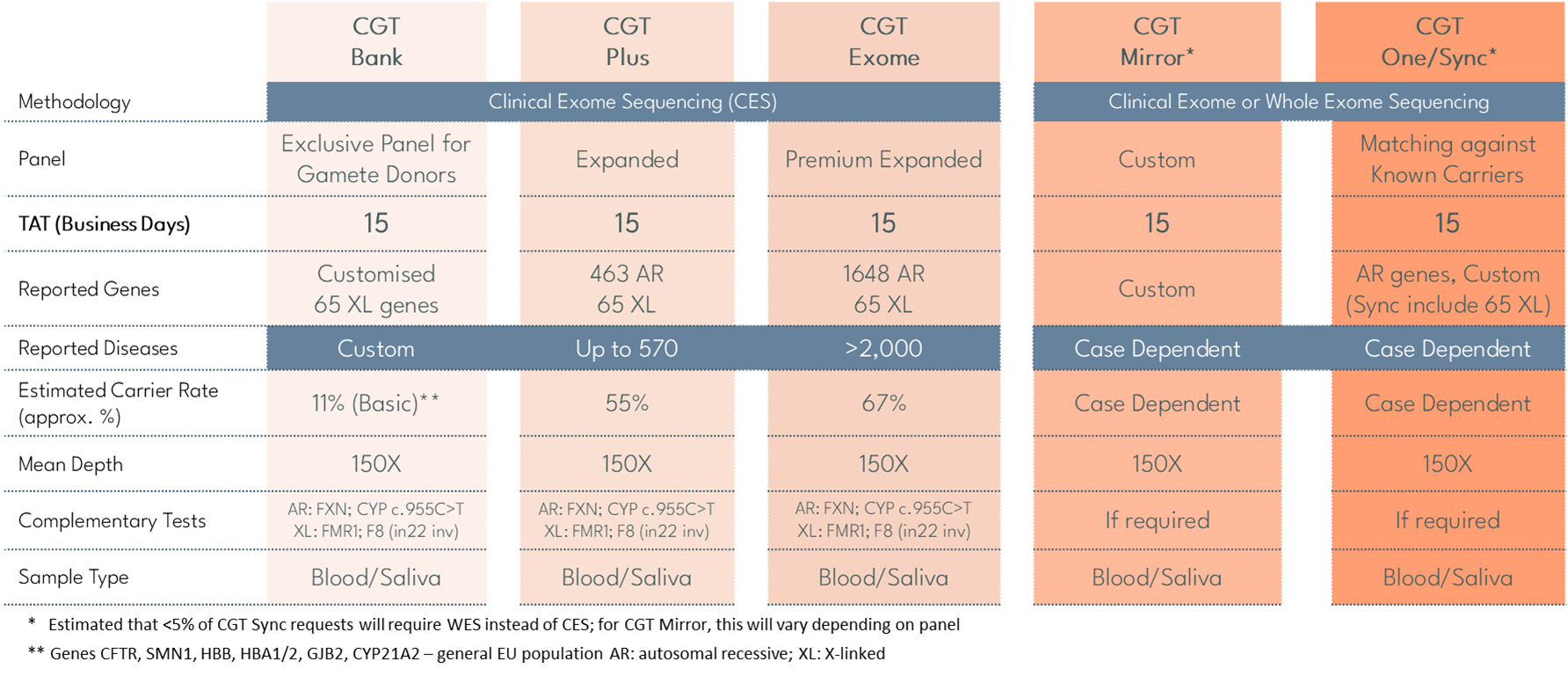
- The Upgraded Carrier Genetic Test (CGT) uses Clinical Exome Sequencing (CES) to identify carrier status in prospective parents for clinically relevant autosomal recessive and X-linked genetic conditions that may be transmitted to offspring.
- Upgraded CGT analyses a broad panel of genes associated with conditions such as Cystic Fibrosis (CF), Spinal Muscular Atrophy (SMA) and Fragile X Syndrome, supporting clinically focused carrier screening and preconception risk detection.
Our New Universal approach to expanded carrier screening using Clinical Exome Sequencing

CGT Exome
Premium expanded panel for a comprehensive analysis with maximum detection.

CGT Plus
Expanded panel aligned with clinical recommendations.

CGT Bank
Optimised for donor screening and reuse of genetic data for universal matching.
More genes analysed provides higher probability of identifying carrier status
Expanded panels increase the likelihood of detecting relevant genetic risk and improve matching accuracy between partners or donors.
Matching
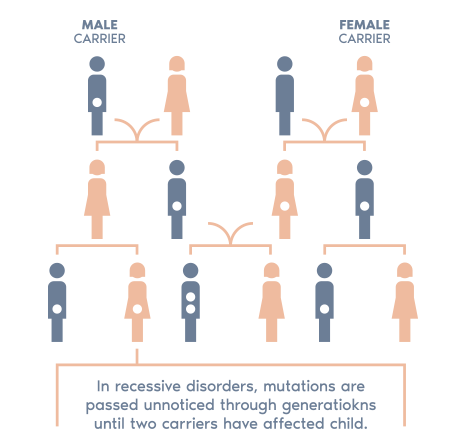
Why testing both partners matters
- Carrier screening is most informative when both partners are tested. If both carry the same condition, there is an increased potential risk to their future child
- Our approach supports efficient matching between partners or between individuals using donors
- The test is prescribed by the doctor
- A sample of either blood or saliva is collected
- DNA is analysed using advanced sequencing technology
- Results are reported to support your reproductive decisions